While the current brain-computer interface (BCI) landscape is defined by motor recovery for the paralyzed, a new frontier is emerging. While giants like Neuralink, Synchron, and Paradromics focus on digital telepathy, a new brain implant for depression is moving into human clinical trials. Houston-based startup Motif Neurotech has recently received FDA approval to begin testing its device designed to combat severe mental health disorders.
How This Brain Implant for Depression Targets the Central Executive Network
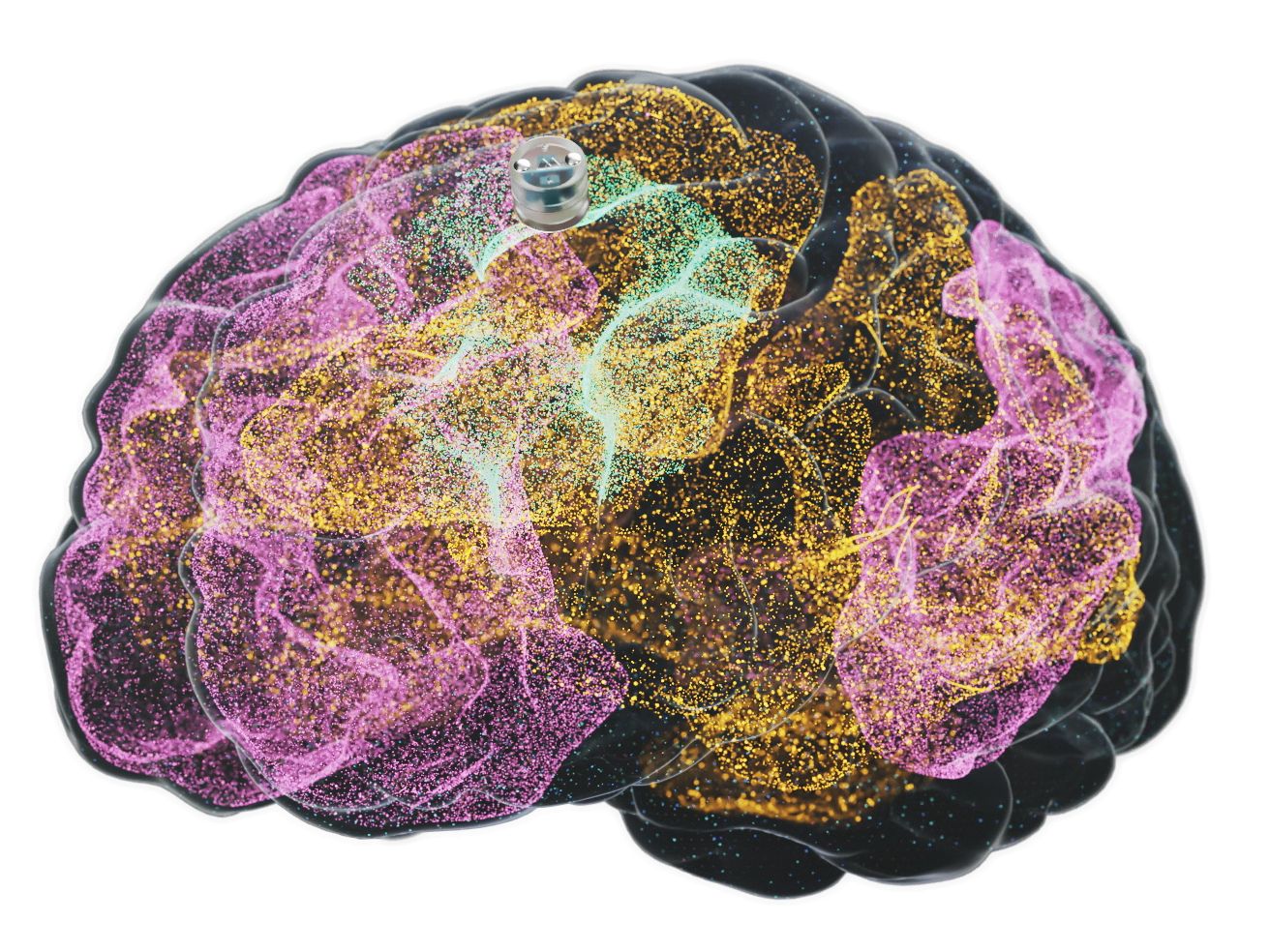
The technology consists of a blueberry-sized stimulator positioned just above the dura, the brain's protective membrane. Unlike invasive deep-brain procedures, Motif’s device utilizes wireless magnetoelectric technology to deliver electrical stimulation. It specifically targets the central executive network, a region responsible for high-level cognitive functions.
This specific neural network has been identified as chronically underactive in individuals suffering from major depressive disorder. By using precise stimulation patterns, the device aims to "turn on" this network. The ultimate goal is to drive neuroplasticity, allowing the brain to reorganize itself by forming new, healthier neural connections.
Bridging the Gap in Neuromodulation
For decades, clinicians have relied on several methods to treat resistant depression, but each carries significant burdens:
- Electroconvulsive Therapy (ECT): A high-intensity intervention involving controlled seizures.
- Deep Brain Stimulation (DBS): Highly invasive surgery requiring implanted electrodes.
- Transcranial Magnetic Stimulation (TMS): A non-invasive approach that requires a grueling schedule of five treatments per week.
Motif Neurotech seeks to bridge the gap between these methods. The implantation is a simple, 20-minute outpatient procedure that avoids traditional brain surgery. Furthermore, the device is designed for at-home use via a specialized baseball cap that delivers prescribed doses of stimulation.
The Path to Objective Psychiatry
The upcoming 12-month clinical trial will enroll approximately ten participants to evaluate the safety and efficacy of the device. While the primary focus remains on safety, researchers will monitor for reductions in anxiety, depressive symptoms, and improved cognitive function. This study represents a massive leap toward objective psychiatry.
Currently, psychiatric diagnosis relies on subjective patient reporting and clinician observation. The long-term vision for this technology involves using the implant to record and monitor brain activity over time. This longitudinal data could move physicians away from "trial and error" prescribing.
By utilizing real-time, measurable changes in neural signaling, the medical field can transition toward personalized neurostimulation. If successful, this shift could transform depression from a condition managed by guesswork into one treated with the precision of digital medicine.